CHOLBAM has been shown to improve liver function parameters and support weight gain in patients with PBD‑ZSD with liver involvement1
Results of 2 trials, including an 18-year open-label trial and
a 21-month extension, of patients with PBD‑ZSD (N=24)¹,†
| Peroxisomal Disorder | Responders/ number treated (%) |
|---|---|
| Zellweger syndrome | 3/8 (38%) |
| Neonatal adrenoleukodystrophy | 3/6 (50%) |
| Infantile Refsum disease | 3/4 (75%) |
Two additional responders are not shown; 1 had generalized peroxisomal disorder and 1 had an unknown peroxisomal disorder.¹
46% (11/24) of evaluable patients responded to CHOLBAM treatment.1,‡

38% of the responders met the 2 clinical criteria plus ≥1 of the following laboratory criteria1:
- ALT or AST values reduced to less than 50 U/L, or baseline levels reduced by 80%.
- Total bilirubin values reduced to ≤1 mg/dL.
- No evidence of cholestasis on liver biopsy was observed.

63% of responders (7/11) showed improved weight gain1
- Body weight increased by 10% or stabilized at >50th percentile.
Response criteria: Response to CHOLBAM treatment was assessed using the following criteria1:
Laboratory criteria: (1) ALT or AST values reduced to <50 U/L, or baseline levels reduced by 80%; (2) total bilirubin values reduced to ≤1 mg/dL; and (3) no evidence of cholestasis on liver biopsy.
Clinical criteria: (1) body weight increased by 10% or stable at >50th percentile; (2) survival for >3 years on treatment or alive at the end of Trial 2.
CHOLBAM responders were defined as patients who either: (1) met at least 2 laboratory criteria and were alive at last follow-up or (2) met at least 1 laboratory criterion, had increased body weight, and were alive at last follow‑up.1
†Twenty-nine patients with PBD-ZSD were treated over an 18-year period in a nonrandomized, open-label, single-arm trial. Twelve patients were treated in an extension trial (2 new patients and 10 patients who rolled over from the original trial). Efficacy data are available for 21 months of treatment in the extension trial.1
‡Sufficient data were available to assess baseline liver function and effects of CHOLBAM treatment in 23 patients in the original trial and in 1 new patient in the extension trial.1
ALT, alanine aminotransferase; AST, aspartate aminotransferase.
42% of patients (13/31) survived >3 years from the time of trial entry1
- 62% (8/13) of these patients survived on treatment from 10 to 17 years.
- No evidence of improvement in survival compared with that seen in historical controls could be demonstrated from the data presented.
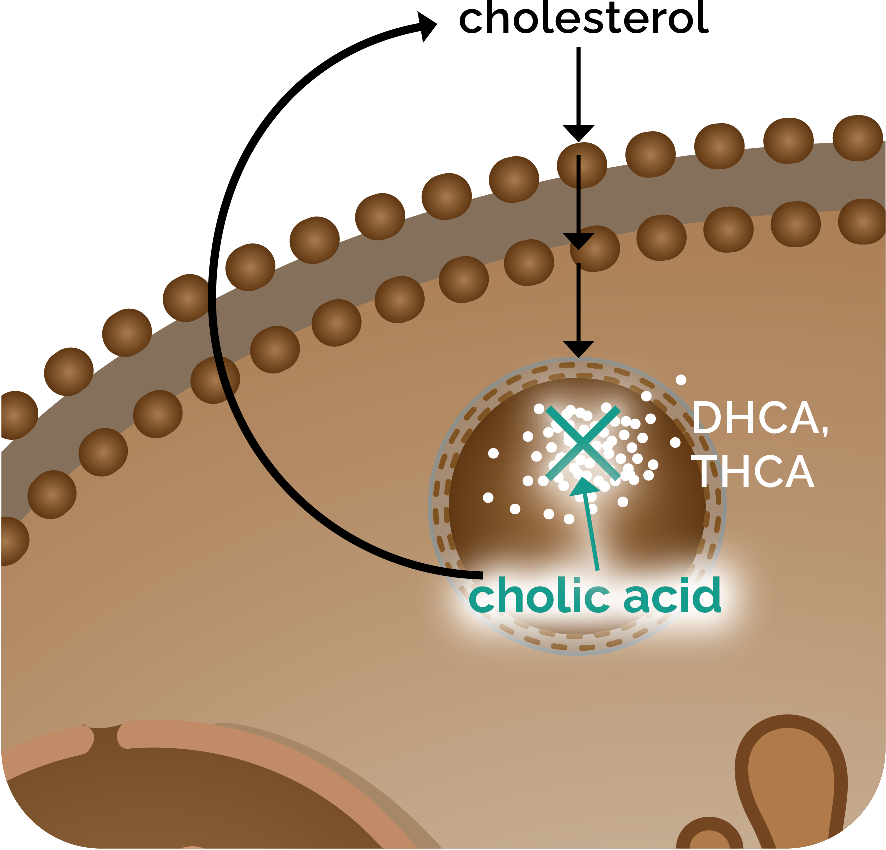
CHOLBAM reduces toxic accumulation of atypical bile acids3
CHOLBAM provided a significant decrease in atypical bile acids in patients with PBD‑ZSD without advanced liver disease.3
In an open-label study of 19 patients living with PBD‑ZSD, CHOLBAM 15 mg/kg/day was administered to 15 patients without advanced liver disease and 4 patients with severe fibrosis or cirrhosis for a period of 9 months. The dosage was increased to 20 mg/kg/day if C27-bile acid intermediates dihydroxycholestanoic acid (DHCA) and/or trihydroxycholestanoic acid (THCA) were still detectable in plasma. In case of clinical side effects, particularly diarrhea, vomiting, or biochemical effects defined as a 2-fold increase in plasma transaminases or conjugated bilirubin, the dosage was reduced to 10 mg/kg/day.3,§,||
The effect of CHOLBAM on plasma DHCA and THCA in patients
with PBD-ZSDs without advanced liver disease (n=15)3,a
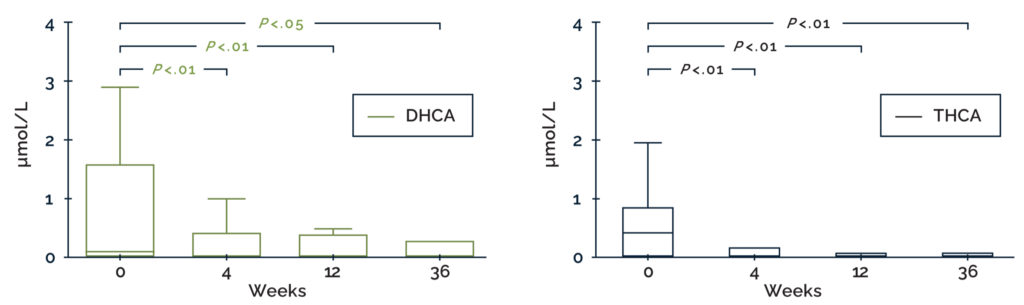
Box-and-whisker plots showing the effect of CHOLBAM therapy on atypical bile acid levels. Patients were followed for 2 to 2.5 years prior to the start of intervention with CHOLBAM. The reference range for THCA is <0.05–0.1 µmol/L; DHCA is undetectable in controls (<0.05 µmol/L). Statistical analyses were performed using a Wilcoxon matched-pairs signed-rank sum test. aOutliers have been omitted from the graph.
Adapted from Berendse K et al, J Inherit Metab Dis, 2016.
- 31% of patients (4/13) with elevated levels of DHCA/THCA at baseline had undetectable atypical bile acids after 9 months of CHOLBAM treatment.3
- Patients with advanced liver disease exhibited persistently elevated atypical bile acid levels during cholic acid treatment.3
§Liver stiffness was evaluated using ultrasound-based transient elastography (FibroScan®) and scored according to the Metavir fibrosis scoring system for chronic cholestatic liver disease. FibroScan values ≥15.5 kPa were defined as severe fibrosis or cirrhosis.3
||Please note that the recommended dosage is 10 to 15 mg/kg/day.1 Refer to full Prescribing Information for full dosage and administration information.
CHOLBAM has a well-established safety profile
The safety and effectiveness of CHOLBAM have been demonstrated with more than 18 years of clinical trial experience.1
There were 12 adverse reactions reported across 9 patients in the trials.1
Most common adverse reactions in trials 1 and 21
| Adverse reactions | Trial 1 | Trial 2# | Overall (%) |
| Diarrhea | 1 | 2# | 3 (2%) |
| Reflux esophagitis | 1 | 0 | 1 (1%) |
| Malaise | 1 | 0 | 1 (1%) |
| Jaundice | 1 | 0 | 1 (1%) |
| Skin lesion | 1 | 0 | 1 (1%) |
| Nausea | 0 | 1# | 1 (1%) |
| Abdominal pain | 0 | 1# | 1 (1%) |
| Intestinal polyp | 0 | 1# | 1 (1%) |
| Urinary tract infection | 0 | 1# | 1 (1%) |
| Peripheral neuropathy | 0 | 1 | 1 (1%) |
#Adverse reactions that occurred in new patients.

Learn about no-cost tests for your PBD‑ZSD patients

Get your PBD‑ZSD patients
started on CHOLBAM
REFERENCES: 1. CHOLBAM. Prescribing information. Mirum Pharmaceuticals, Inc. 2. Heubi JE, Setchell KDR, Bove KE. Long-term cholic acid therapy in Zellweger spectrum disorders. Case Rep Gastroenterol. 2018;12(2):360-372. doi: 10.1159/000490095. 3. Berendse K, Klouwer FCC, Koot BGP, et al. Cholic acid therapy in Zellweger spectrum disorders. J Inherit Metab Dis. 2016;39(6):859-868. doi: 10.1007/s10545-016-9962-9. 4. Braverman NE, Raymond GV, Rizzo WB, et al. Peroxisome biogenesis disorders in the Zellweger spectrum: An overview of current diagnosis, clinical manifestations, and treatment guidelines. Mol Genet Metab. 2016;117(3):313-321. doi: 10.1016/j.ymgme.2015.12.009. 5. Zeynelabidin S, Klouwer FCC, Meijers JCM, et al. Coagulopathy in Zellweger spectrum disorders: a role for vitamin K. J Inherit Metab Dis. 2018;41(2):249-255. doi: 10.1007/s10545-017-0113-8. 6. Klouwer FCC, Koot BGP, Berendse K, et al. The cholic acid extension study in Zellweger spectrum disorders: Results and implications for therapy. J Inherit Metab Dis. 2019; 42:303–312. doi.org/10.1002/ jimd.12042.
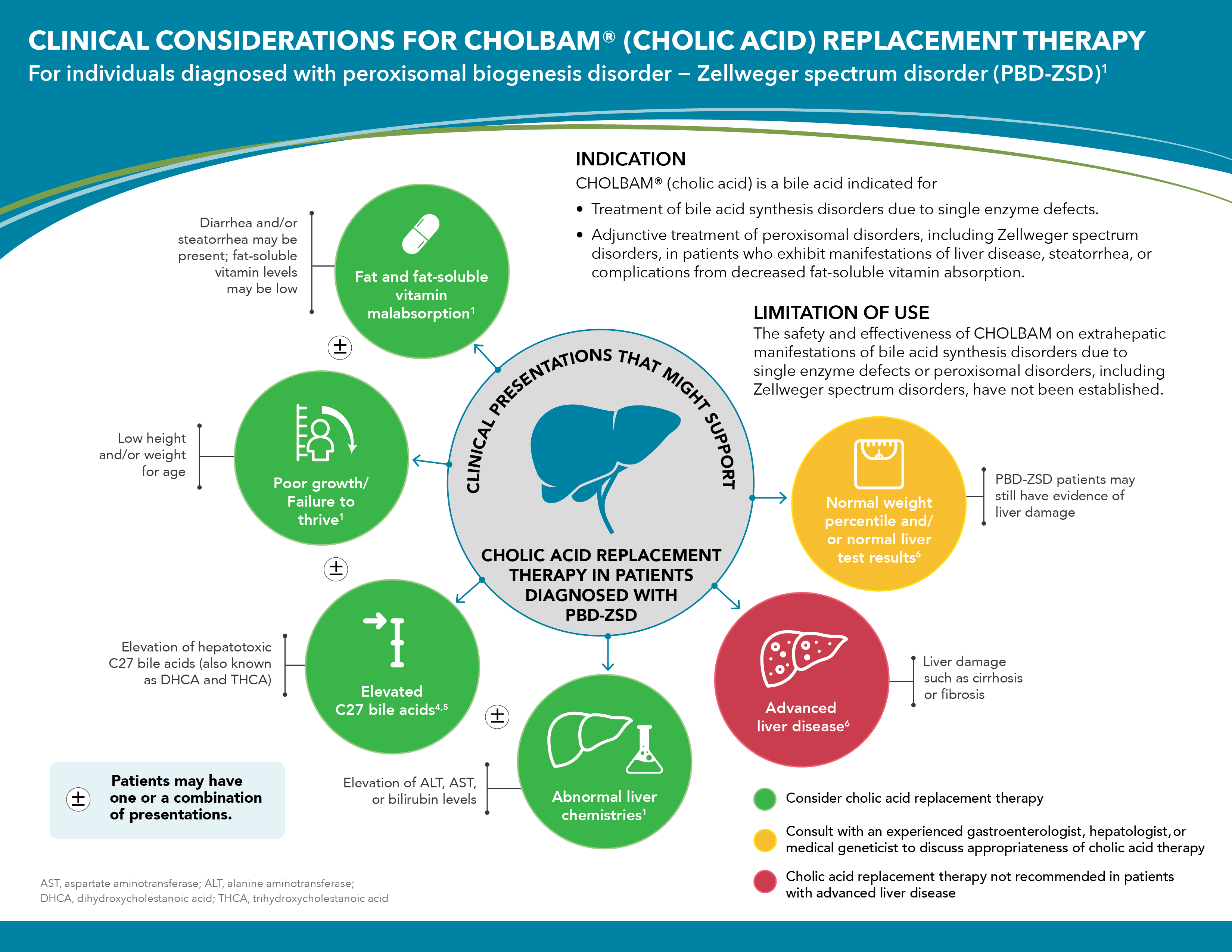
INDICATION
CHOLBAM® (cholic acid) is a bile acid indicated for- Treatment of bile acid synthesis disorders due to single enzyme defects.
- Adjunctive treatment of peroxisomal disorders, including Zellweger spectrum disorders, in patients who exhibit manifestations of liver disease, steatorrhea, or complications from decreased fat-soluble vitamin absorption.
LIMITATIONS OF USE
The safety and effectiveness of CHOLBAM on extrahepatic manifestations of bile acid synthesis disorders due to single enzyme defects or peroxisomal disorders, including Zellweger spectrum disorders, have not been established.IMPORTANT SAFETY INFORMATION
WARNINGS AND PRECAUTIONS – Exacerbation of liver impairment
- Monitor liver function and discontinue CHOLBAM in patients who develop worsening of liver function while on treatment.
- Concurrent elevations of serum gamma glutamyltransferase (GGT) and alanine aminotransferase (ALT) may indicate CHOLBAM overdose.
- Discontinue treatment with CHOLBAM at any time if there are clinical or laboratory indicators of worsening liver function or cholestasis.
ADVERSE REACTIONS
- The most common adverse reactions (≥1%) are diarrhea, reflux esophagitis, malaise, jaundice, skin lesion, nausea, abdominal pain, intestinal polyp, urinary tract infection, and peripheral neuropathy.
DRUG INTERACTIONS
- Inhibitors of Bile Acid Transporters: Avoid concomitant use of inhibitors of the bile salt efflux pump (BSEP) such as cyclosporine. Concomitant medications that inhibit canalicular membrane bile acid transporters such as the BSEP may exacerbate accumulation of conjugated bile salts in the liver and result in clinical symptoms. If concomitant use is deemed necessary, monitoring of serum transaminases and bilirubin is recommended.
- Bile Acid Binding Resins: Bile acid binding resins such as cholestyramine, colestipol, or colesevelam adsorb and reduce bile acid absorption and may reduce the efficacy of CHOLBAM. Take CHOLBAM at least 1 hour before or 4 to 6 hours (or at as great an interval as possible) after a bile acid binding resin.
- Aluminum-Based Antacids: Aluminum-based antacids have been shown to adsorb bile acids in vitro and can reduce the bioavailability of CHOLBAM. Take CHOLBAM at least 1 hour before or 4 to 6 hours (or at as great an interval as possible) after an aluminum-based antacid.
PREGNANCY
No studies in pregnant women or animal reproduction studies have been conducted with CHOLBAM.LACTATION
Endogenous cholic acid is present in human milk. Clinical lactation studies have not been conducted to assess the presence of CHOLBAM in human milk, the effects of CHOLBAM on the breastfed infant, or the effects of CHOLBAM on milk production. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for CHOLBAM and any potential adverse effects on the breastfed infant from CHOLBAM or from the underlying maternal condition.GERIATRIC USE
It is not known if elderly patients respond differently from younger patients.HEPATIC IMPAIRMENT
- Discontinue treatment with CHOLBAM if liver function does not improve within 3 months of the start of treatment.
- Discontinue treatment with CHOLBAM at any time if there are clinical or laboratory indicators of worsening liver function or cholestasis. Continue to monitor laboratory parameters of liver function and consider restarting at a lower dose when the parameters return to baseline.